Silicone radial head prostheses revisited: do they have a role in today’s practice? A systematic review of literature on clinical outcomes
Article information
Abstract
Background
Silicone radial head prostheses (SRHP) are considered obsolete due to reports of frequent failure and destructive silicone-induced synovitis. Considering the good outcomes of modern non-radial silicone joint implants, the extent of scientific evidence for this negative view is unclear. The aim of this research was to systematically analyze the clinical evidence on complications and outcomes of SRHP and how SRHP compare to both non-SRHP and silicone prostheses of other joints.
Methods
A systematic literature review was conducted through the Cochrane, PubMed, and Embase databases.
Results
Eight cohort studies were included, consisting of 142 patients and follow-up periods ranging from 23 months to 8 years. Average patient satisfaction was 86%, range of 71%–100%, and 58 complications were seen, but no cases of synovitis. These outcomes were in line with non-SRHP. Four case series with 11 cases of synovitis were found, all due to implant fractures years to decades after implantation. Six systematic reviews of currently used non-radial silicone joint implants showed excellent outcomes with low complication rates.
Conclusions
Since SRHP have satisfactory clinical results and an acceptable complication rate when selecting a patient group in suitable condition for surgical indications, it is considered that SRHP can still be chosen as a potential surgical treatment method in current clinical practice.
Level of evidence
2A.
INTRODUCTION
The biomechanical importance of the radial head has been studied extensively over recent decades. The radial head plays a role in tensioning the lateral collateral ligament and provides axial stability of the forearm together with the interosseous membrane [1]. The radial head also serves as the secondary valgus stabilizer in addition to the medial ulnar collateral ligament [2,3]. Due to its significant contribution to stability, preservation of the radial head is considered essential in fracture treatment [4]. However, in cases of irreparable traumatic destruction, the radial head is removed or replaced by a prosthesis. Radial head replacement with a prosthesis has been an acceptable option when associated ligament injury is present [5,6].
Over the past six decades, several designs of radial head prostheses (RHP) have been developed [5,6]. Designs differ in biomechanical properties, such as modularity, head size and connection to the stem, polarity, methods of fixation, and materials used [5,7]. One of the designs was the silicone radial head prosthesis (SRHP), which was widely used during the 1970s and 1980s. However, these prostheses are now considered obsolete in elbow surgery based on a number of reports that were published decades ago. They showed clinical failure as a result of mechanical failure and so-called “silicone-induced synovitis due to silicone debris” [8-11].
However, silicone implants are still widely used for replacements in the wrist, hand joints, and forefoot [12,13]. The design and mechanical properties of these silicone implants have been improved by advancements in materials such as in tensile strength or tear resistance, resulting in more durable implants with less mechanical failure and excellent clinical outcomes [13-16].
The development of non-silicone RHPs has continued as well, resulting in prostheses of various designs and materials. However, revision and complication rates are still high, up to 23% according to the review by Heijink et al. [6]. It is quite unusual that SRHP fell into complete disuse, while usage of silicones in other joints was continued. Also, the two most recent papers concerning a cohort of the latest design SRHP did not show any cases of revision due to implant failure among their 43 cases [17,18].
This divergence in usage of silicones in joint implants could raise the question as to how inferior in function the SRHP actually was. More specifically, how would complication rates of SRHP relate to both outcomes of non-silicone RHP and to outcomes of silicone implants in other joints. Unfortunately, no systematic review of the literature concerning SRHP has been published to date. Two studies, published in 2011 and 2013, showed that SRHP can have excellent outcomes as long as certain conditions such as ligamental stability or temporary usage were taken into consideration [17,18]. Considering the good outcomes of modern non-radial silicone joint implants, the high complication rates of modern non-SRHP, and the acceptable outcomes of SRHP in these individual studies, the extent of the scientific evidence on which SRHP fell into disuse is unclear.
In this study, we sought to (1) systematically review the available literature on outcomes of silicone RHPs, (2) systematically review the available literature on outcomes of silicone implants in other joints, and (3) put the outcomes of SRHP in perspective compared to non-silicone RHP and implants in other joints.
METHODS
This systematic review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist criteria and guidelines [19]. At the start of this study, the review was registered at PROSPERO, the international prospective register of systematic reviews, with ID 218202. Since the aim of this study was binominal, two searches were performed. First, all studies concerning SRHP were analyzed. Second, all systematic reviews concerning non-radial silicone arthroplasties were evaluated. The searches were performed on December 1, 2021.
Silicone Radial Head Prostheses
Study outcome and search strategy
All relevant studies concerning outcomes of SRHP were analyzed. Due to the limited number of published studies regarding SRHP, as many studies as possible were included as long as the following inclusion criteria were met: (1) a clear description of the total number of studied SRHP, (2) a clear description of the follow-up, and (3) a description of the clinical outcome and complication rate of all included SRHP. Due to these broad inclusion criteria, it was decided not to pool the data.
No exclusion criteria were stated concerning year of publication or type of study, other than exclusion of case reports and articles with non-primary content (e.g., theses, expert opinions, letters to the editor). Titles were excluded if no full article was available or if the study design and method of data analysis were not clearly described. Studies were excluded if the full-text was written in a language other than English, German, French, or Dutch.
The first step in the search process was to find potentially relevant systematic reviews, published and unpublished, by searching the Cochrane database and the PROPSPERO international prospective register of systematic reviews. Second, the databases of www.clinicaltrials.gov and the World Health Organization trial search (https://apps.who.int/trialsearch) were screened to detect current ongoing clinical studies concerning SRHP. Lastly, a comprehensive literature search was performed in Medline via PubMed and Embase with the assistance of a clinical librarian.
Every search consisted of a combination of the following terms: “radial head” or “radial fracture” or 𠇜elbow” and “prosthesis” and“ silicone.” Synonyms and closely related terms were included, such as ”arthroplasty” and ”silastic’.” No filters were applied concerning publication date, language, study design, or level of evidence. Two researchers (EMB and ALL) independently assessed all titles and abstracts, after which the full-texts of all potentially eligible abstracts were read. When there was disagreement with respect to study selection, a third researcher was involved to make the final decision.
Study selection, quality assessment, and data analysis
All selected studies were assessed for their quality. Since mainly qualitative (retrospective) studies were expected, the “Critical Appraisal Skills Programme” or “CASP” checklist for qualitative studies was used, comprising ten criteria in terms of validity, method used, outcome, and usefulness. Studies were considered as “high quality” if at least 8 of the 10 criteria were met, as “medium quality” if 5–7 criteria were met, or as “low quality” for meeting fewer than 5 criteria [20,21].
Selected studies were systematically analyzed, in which the following data were gathered and described: (1) number of patients and mean follow-up, (2) main subject of the article, (3) clinical and functional outcomes, and (4) complications including synovitis, failure/revision/removal rates, and radiological assessments. Due to the expected heterogenic and qualitative nature of the selected studies, only a limited statistical analysis was performed, with the only goal being to put studies in perspective.
Systematic Reviews of Silicone Arthroplasties in Other Joints
Study outcome and search strategy
All systematic reviews focusing on clinical outcomes of non-radial silicone arthroplasties were gathered and assessed using the Cochrane, PubMed, and Embase databases. In the searches, the terms “prostheses” or “joint,” and “silicone” were used, including synonyms and closely related terms. The limitation “review” was added as a search term or set as a filter, together with a publication date from the year 2000 onwards.
Study selection and analysis
Two researchers again independently assessed the systematic reviews. Articles were excluded if no systematic literature search was used or if outcomes were only published with pooled data of implants from other materials than silicones. Outcome and complications were gathered and described per systematic review.
RESULTS
Silicone Radial Head Prostheses
Study selection
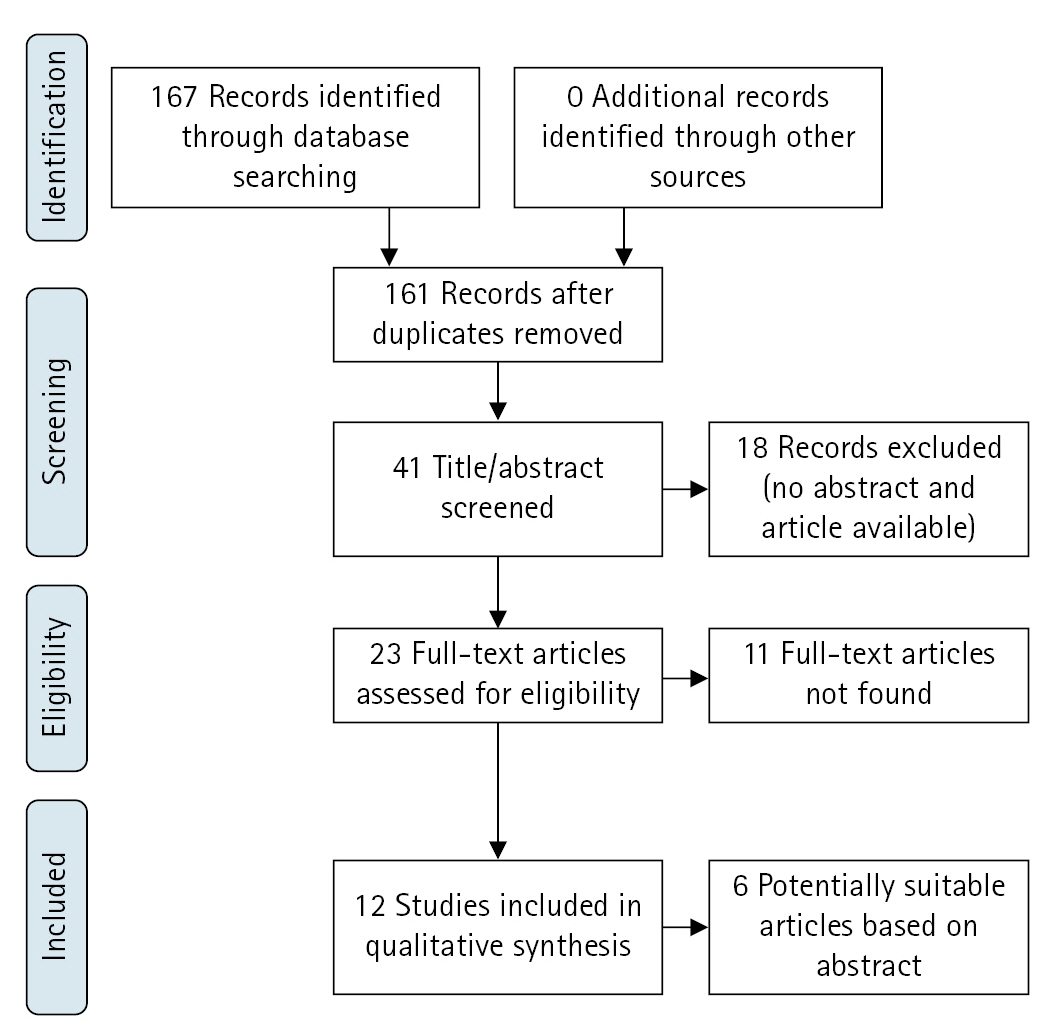
A total of 167 published articles were identified, of which 162 articles were available for screening of title and abstract. No systematic review, published or registered, and no ongoing clinical studies were found. A total of 41 articles were identified as potentially eligible. Reference checking resulted in no additional articles. The process of study selection is shown in the flowchart in Fig. 1 [19].
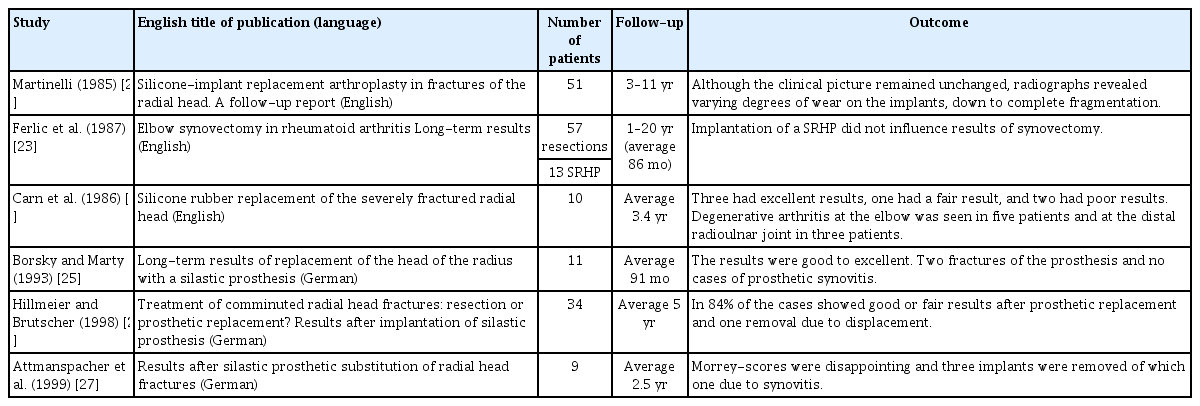
For 18 results, all in non-English languages, no abstract or original article could be found. For 11 results, all written before 2000, the abstract was available, but no article was found. Nevertheless, five of these abstracts did describe the majority of the stated outcome criteria, such as the number of studied SRHP, notation of the follow-up, and suggestion of clinical outcomes of SRHP. These untraceable, potentially eligible articles are described in Table 1 [22-27]. Twelve primary articles, consisting of 153 SRHP, were found and considered suitable for further qualitative analysis [8,11,13,24,25,28-34]. These studies ranged from 1979 until 2019, with four articles being less than 10 years old [17,18,28,29].
Study characteristics and quality assessment
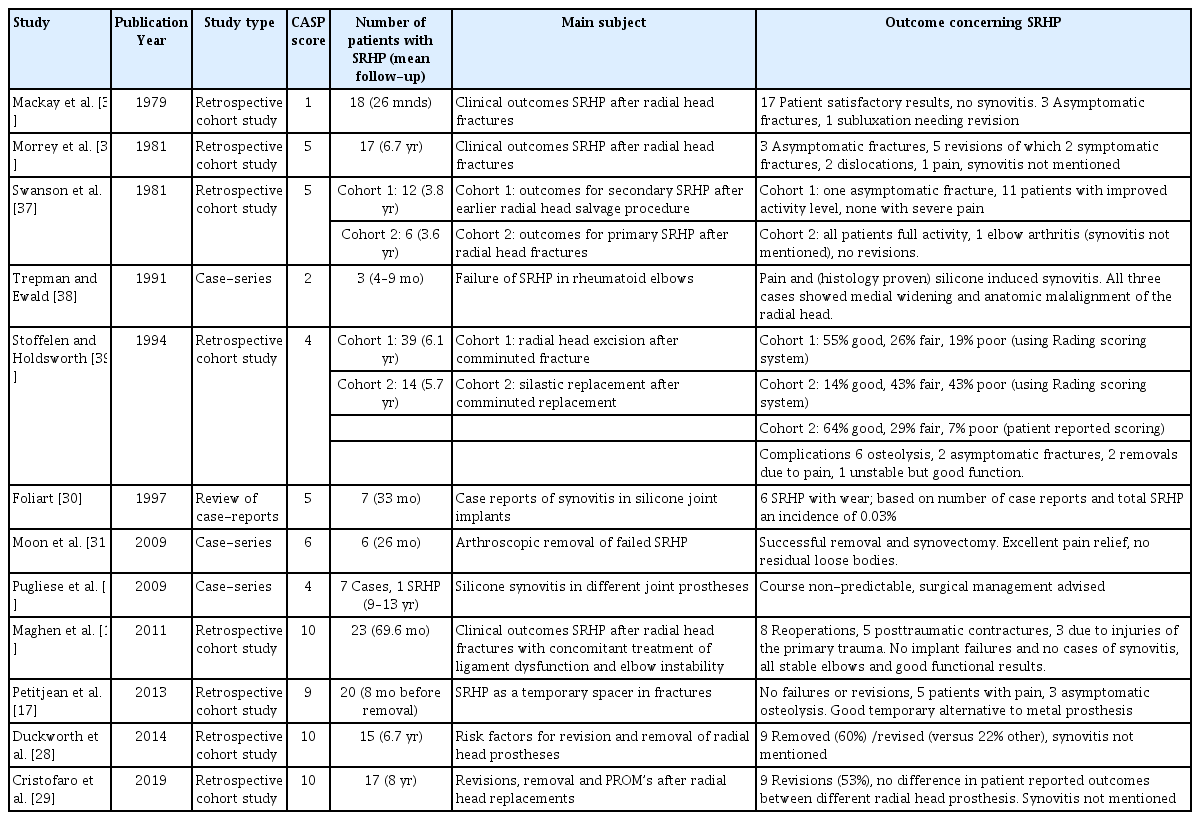
All twelve selected studies, eight of which were cohort studies and the other four being case series, were retrospective studies (Table 2) [17,18,29-31,35-40]. Within the eight retrospective cohort studies, six focused solely on SRHP, and two compared SRHP with radial head excision [17,18,28,29,35-37,39]. Furthermore, SRHP were used as a permanent prosthesis in seven cohort studies and as a temporary spacer in one cohort [17]. Two cohort studies, also being the two most recent publications, analyzed risk factors for removal and revision as their main outcome, in which SRHP was one of the studied factors [28,29]. Of the four case series, two studies described a series of silicone synovitis in different silicone joint prostheses, including SRHP [30,40]. One case series documented a series of clinically failed SRHP in rheumatoid elbows, and one case series reported arthroscopic removal of failed SRHP [31,38].
Using the quality assessment of the CASP checklist for qualitative studies, the quality of four studies was categorized as low and that of four studies as medium (Table 2). The quality of the remaining four studies was categorized as high; these were the four most recently published studies as well, with dates of publication ranging from 2011 to 2019 [17,18,28,29].
Outcomes of SRHP
The study populations of silicone RHPs ranged from 1 to 23 cases among the 12 selected studies. Within the eight cohort studies, the range of the individual cohorts was 6 to 23 elbows with a mean follow-up ranging from 23 months to 8 years (Table 2) [17,18,28,29-31,35-40].
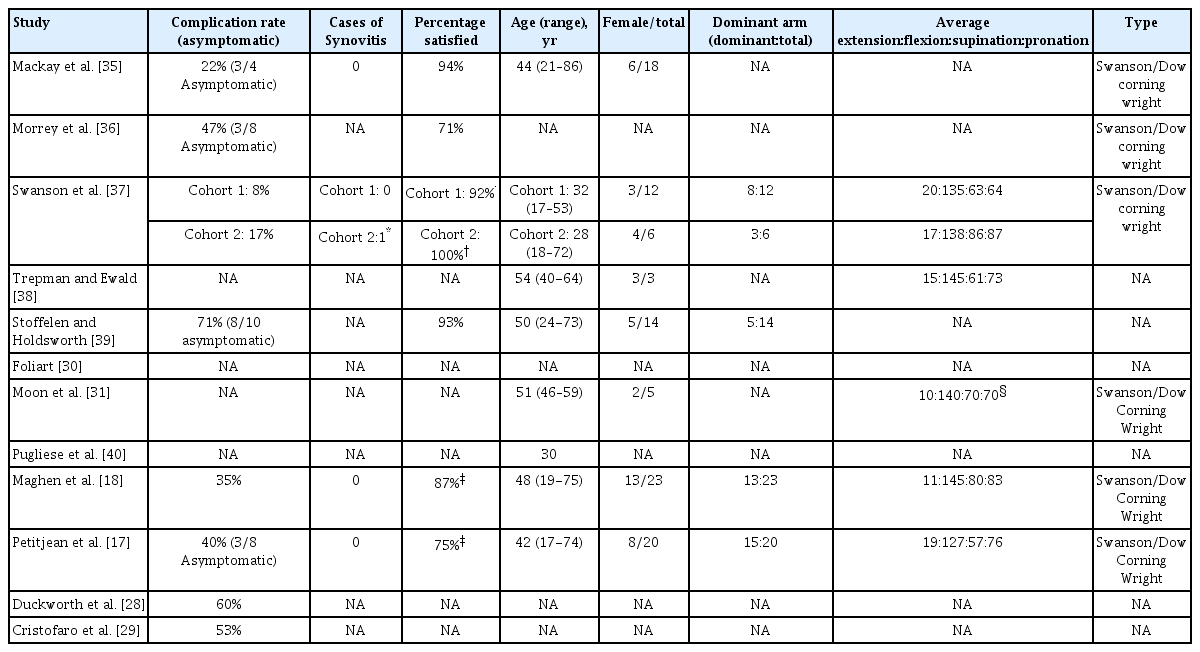
There were a total of 142 patients in the cohort studies that had a total of 58 complications (41%). It was unclear how many patients had more than one complication. The complication rates within these cohorts ranged from 8% to 71% (Table 3) [37,39]. Seventeen complications were asymptomatic, consisting of radiographic fractures of the prosthesis (n=8) and osteolysis (n=9) [17,35,36,39]. If one were to exclude these asymptomatic complications, the complication rate would range from 0 to 60% among the cohorts, with an average complication rate of 29%. For the two most recent cohort studies, no direct or indirect measure of patient satisfaction was reported [28,29]. In the other six cohort studies, the mean patient satisfaction level was 86% and ranged from 71% to 100% after an average of 49 months (Table 3) [18,35,36,39]. Functional scores were measured in three studies, although with different scoring systems [28,29,27].
Four cohort studies reported no cases of silicone synovitis, whereas Swanson et al. [37] did describe one patient with elbow arthritis. Other than an intact implant and no need for revision, no further details, such as a suspected septic origin or treatment, were described for this case. For the remaining three cohort studies, the incidence of silicone synovitis was not described [28,29,39]. It is uncertain whether this means that no cases were observed or that assessments tools were not sufficient.
In addition to the eight cohort studies, four case series were found in this systematic review. In these four case series, 11 cases of synovitis in SRHP are described [30,31,38,40]. This differs from the documentation of only a single case among the 142 patients within the cohort studies. Not all four case series focus solely on SRHP. The case series by both Foliart [30] and Pugliese et al. [40] describe synovitis in different silicone joint prostheses. Foliart [30] describe a total of 182 cases of silicone-induced synovitis, including seven of the radial head, based on published case reports and case series. The estimated incidence of synovitis for SRHP in this study is 0.03%, although the statistical substantiation of this value may be limited. The study of Pugliese et al. [40] describes seven cases of synovitis, one of which concerns SRHP. This study does not provide any further quantitative results but does demonstrate that synovitis can be induced long after implantation (range, 9–30 years). Five of the seven joint implants in this study were surgically removed. In all cases, the implant was fractured. Together with a foreign body type reaction found in histology, the authors suggest a correlation between this fracturing and a consequential foreign body-induced synovitis.
The third case series of Trepman and Ewald [38] describe three cases of silicone-induced synovitis of an elbow in patients with an SRHP and suffering from rheumatoid arthritis. Remarkably, failure was already seen after 4 to 9 months of implantation. All three cases were clinically, radiologically, and histologically analyzed. Although this study does not add much value in a quantitative sense, all three cases showed a fractured or frayed silicone prosthesis and a histologically foreign body type immune response during analysis, which was comparable to the study by Pugliese et al. [40]. In the last case series by Moon et al. [31], a surgical technique for arthroscopic removal is described. Despite the different focus of this paper, the study also demonstrates fracturing of the silicone implants in all cases and clinical failure after a period of 2 to 18 years after implantation.
Next to the four included case series, six abstracts were found that were potentially eligible but of which no full-text could be retrieved [22-27]. Although conclusions based on these abstracts are of limited value, the results seem to be in line with those of the other outcomes: a clinically well tolerated implant, with varying complication rates but a limited number of synovitis cases (Table 1).
Systematic Reviews of Silicone Arthroplasties in Other Joints
Selection of systematic reviews and characteristics
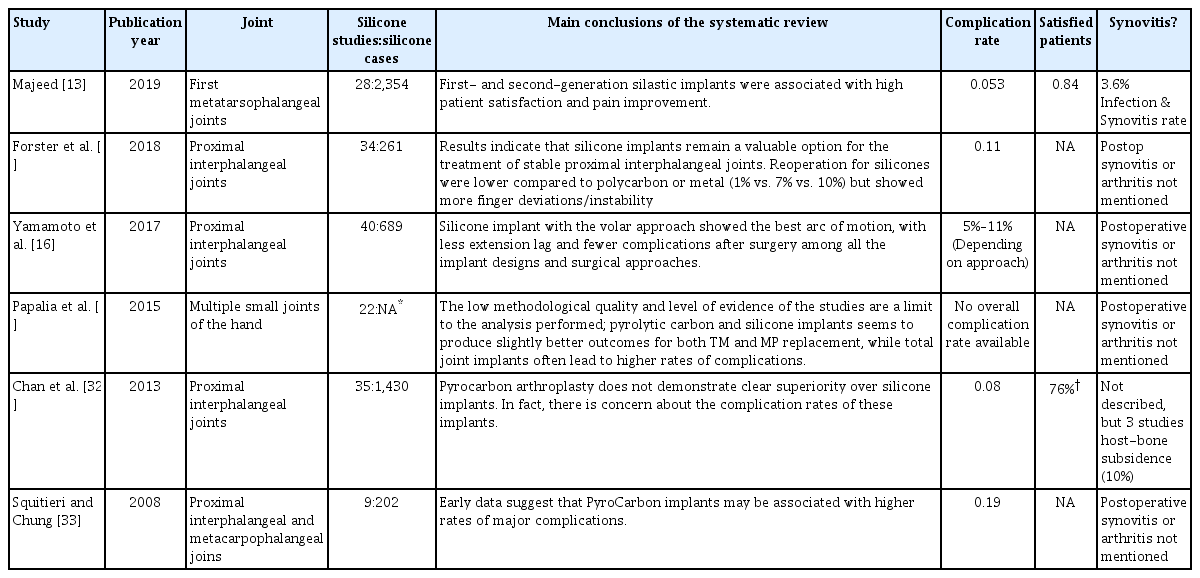
Nineteen potentially eligible reviews were found. Full-text were available for 18 reviews. No Cochrane reviews or ongoing systematic reviews were found. Six articles, published between 2008 and 2019, reviewed the available literature systematically and were further evaluated (Table 4) [12,13,15,16,32,33].
One systematic review concerned a silicone joint implant in the lower extremity (first metatarsophalangeal joint) [13]. Five reviews regarded the upper extremity in which the silicone joint prosthesis was compared with carbon arthroplasties. Of these five reviews, three analyzed silicone implants in proximal interphalangeal joints, and two a heterogeneous group of finger joints, mainly the interphalangeal and metacarpal joints. Although these five literature reviews had specific aims, they did have some overlap with their included studies.
Outcomes of systematic reviews
For the six systematic reviews, the analyzed number of studies and silicone prostheses ranged from 9 to 40 studies and from 202 to 2354 cases [12,13,15,16,32,33]. None of the systematic reviews advised against the use of silicone arthroplasties. The two most recent reviews concluded clinical superiority of silicone arthroplasties compared to carbon prostheses [15,16]. In the other three reviews, no outspoken preference was given, although two studies reported concerns about the assumed higher complication rates of carbon implants [32,33].
The highest complication rate, 19%, was seen in the review by Squitieri and Chung (202 cases) [33]. In the systematic review by Majeed [13], being the review with the highest number of analyzed patients (2,354 cases), the complication rate was 5.3%. Patient satisfaction was not clearly described in four of the six reviews. The review by Majeed [13] stated a satisfaction level of 84% (average follow-up of 85.3 months), whereas Chan et al. [32] reported 76% of the patients being pain-free after the silicone joint implantation [31,40].
Postoperative cases of silicone-induced synovitis or arthritis were not reported in five systematic reviews [12,15,32,33]. In the systematic review by Majeed [13] concerning first metatarsophalangeal joint prostheses, an incidence of 3.6% was given for all combined cases of infection or synovitis. In the systematic review by Chan et al. [32], 3 of the 35 included studies described a host vs. bone subsidence of silicone arthroplasties, with an average incidence in these three included studies of 10%. Unfortunately, it was not reported whether this bony subsidence was accompanied by soft tissue inflammation. The reviews included show that silicones can function as a viable and durable material for joint implants.
DISCUSSION
To our knowledge, this is the first systematic review on clinical outcomes of SRHPs. By this systematic analysis, outcomes of SRHP can finally be evaluated based on the available evidence and can be put in perspective compared to non-SRHP and silicone implants in other joints.
The analysis of the literature has led to some interesting findings about SRHP. The results show that the great majority of patients are satisfied with an implanted SRHP. Six studies measuring patient satisfaction showed levels ranging from 71% to 100% [18,35,36,39]. However, among the 142 patients in the eight cohort studies, 58 complications were seen as well [17,18,28-31,35-37,39,40]. Seventeen serious complications, consisting of radiographic fractures of the prosthesis (n=8) and osteolysis (n=9), were perceived as asymptomatic by the patient. This means that radiographic and symptomatic complication rates differed at 41% and 29%, respectively.
Among the 142 patients in the eight cohort studies, only one potential case of silicone-induced arthritis or synovitis was described [37]. This incidence deviates from the 17 cases of silicone-induced synovitis that were described in the four case series [30,31,38,40]. Based on the comprehensive methods and follow-up of the different cohort studies, under-registration in the cohorts does not seem likely. It could therefore be argued either that silicone synovitis occurs mainly after the follow-up time frame of the cohorts (23 months–8 years) or that the actual incidence is rather low. Clues of synovitis due to late material failure were found in the case reports with periods of good functionality for up to 30 years before sudden failure [31,40]. The lack of studies with longer follow-ups and larger cohorts prevents further clarification. This is a shame, since more than 25,000 SRHP were implanted worldwide [30].
The findings should be interpreted with some limitations. Even with broad inclusion criteria to minimize the risk of any selection bias, only 12 suitable studies were found. No prospective or randomized studies were found. The quality of the found studies was variable, with four studies considered of low quality. This limited quality could considerably modify outcomes. None of the cohort studies described ligamental repair or reconstruction in the methodology, although both are known to be important for radial head stability and outcomes of SRHP [4,5,18]. This varying study quality, limited cohort size (142 patients), and a follow-up duration of 23 months to 8 years confine the conclusions concerning the outcomes of SRHP.
Six systematic reviews of silicone implants in joints other than the radial head were found. All these reviews focused on small joint prostheses that are still used in clinical practice [12,13,15,16,32,33]. These reviews show that joint prostheses made of silicones can act as excellent functioning implants with high patient satisfaction (76%–84%) and low complication rates (5.3%–19%). Contrary to SRHP, fracturing, failure, and silicone synovitis do not seem to play a role of great importance. Neither clinical performance nor cost-efficacy seemed inferior to other materials, such as polycarbon [15,33,34].
While the patient satisfaction levels found for SRHP could be called comparable to those of these silicone joint implants, the incidence of clinical complications cannot, with SRHP having a clinical complication rate of 29%. A key contributor for this difference might be found in the different physical properties of the radial head versus smaller joints and in usage of the latest silicone materials with better material characteristics as described by DeCarbo et al. [14]. The combination of high load and inferior material biomechanical properties of silicones is considered to generate this difference in outcomes [7,16, 25,30,34].
However, when outcomes of SRHP are put in perspective to outcomes of non-SRHP, the clinical performances are closer together. A recent review, evaluating all non-SRHP, reported one or more complications in 23% of the included patients and revision rates between 0 to 29% of the included studies, compared to the complication rate of 29% as found in this review [6].
Complications specific to SRHP compared to non-SRHP are osteolysis and silicone-induced synovitis due to material failure [10,40]. This foreign body reaction is not observed for materials used in non-SRHP. However, other complications such as loosening, pain, and stiffness are common for both SRHP and non-SRHP [6,7].
The outcomes of the found literature show that silicones do seem to be a feasible option for joint implants even today, but that the radial head is a demanding location for silicone implants. However, this biomechanical challenge seems to be an ongoing problem for non-silicone RHP.
Due to the excellent functionality of silicone implants in other joints, it can be questioned if there is not still a place for SRHP. Studies by Petitjean et al. [17] and Maghen et al. [18] showed that material failure in SRHP is limited with sufficient (ligamental) stability and that the prosthesis can be used as an easy to place, temporary spacer for complex radial head fractures in an emergency context. Therefore, improved material strength by using upgraded, newer silicone materials with better physical properties and minimizing load of the radial joint by better repair and reconstruction of damaged elbow ligaments may reduce the complication rate and improve outcomes for SRHP [41]. Thus, it would be interesting to examine whether this insight would lead to better results that the cohort studies published more than 25 years ago. Considering the findings of this review, SRHP might be a viable alternative for non-silicone RHP in specific cases. It can therefore be questioned whether this prosthesis was justly removed from clinical use.
This systematic review has shown that the scientific evidence on outcomes of SRHPs is limited. Twelve studies of variable quality were found. The found studies did suggest excellent patient satisfaction and functionality of SRHP in the majority of patients. The character and incidence of complications within different included studies were almost comparable to outcomes of currently used non-SRHP. Recent systematic reviews about silicone implants with the latest design in other joints show excellent results with limited complications and material failure. Together with the satisfactory clinical results and an acceptable complication rate of SRHP, it is considered that SRHP can still be chosen as a surgical treatment method for selective cases in current clinical practice.
Notes
Author contributions
Conceptualization: EMB, ALL.Data curation: EMB, BT. Formal analysis: EMB. Investigation: EMB. Methodology: EMB, ALL. Resources: ALL. Supervision: ALL, BT, DE. Validation: ALL, DE. Writing – original draft: EMB. Writing – review & editing: ALL, BT, DE.
Conflict of interest
None.
Funding
None.
Data availability
None.
Conflict of interest
None.
Acknowledgments
None.