 |
 |
- Search
| Clin Shoulder Elb > Volume 26(4); 2023 > Article |
|
Abstract
Background
Methods
Results
NOTES
Author contributions
Conceptualization: PA, PM, ABH, JB, PL, RM, MJB. Data curation: PM, ABH, JB, PL, RM, MJB. Formal analysis: PA, ABH, JB, PL, EGM. Funding acquisition: EGM. Investigation: PA, MJB, EGM. Methodology: PA, PM, ABH, JB, PL, RM, MJB. Project administration: RM. Resources: PM, ABH, JB, PL. Supervision: PM, ABH, PL, MJB. Validation: PM, PL, MJB, EGM. Visualization: EGM. Writing – original draft: PA, PM, ABH, JB, PL, RM, MJB, EGM. Writing – review & editing: PA, PM, ABH, JB, PL, RM, MJB, EGM.
Conflict of interest
EGM is a consultant for Stryker Corporation, outside of this work. All authors declare that they, their immediate family, and any research foundation with which they are affiliated received no financial payments or other benefits from any commercial entity related to the subject of this article.
Funding
The authors disclose receipt of the following financial or material support for the research, authorship, and/or publication of this article: The Donna and William Marriott Research Fund. This funding source had no role in the design, execution, analyses, interpretation of the data, or decision to submit results.
Acknowledgments
For editorial assistance, we thank Denise Di Salvo, MS, and Rachel Walden, MS, in the Editorial Services group of the Johns Hopkins Department of Orthopedic Surgery. We also thank the senior author's physician assistant, Marcy Beard, PA-C, Johns Hopkins Medicine, for her involvement in patient examinations.
Fig. 1.
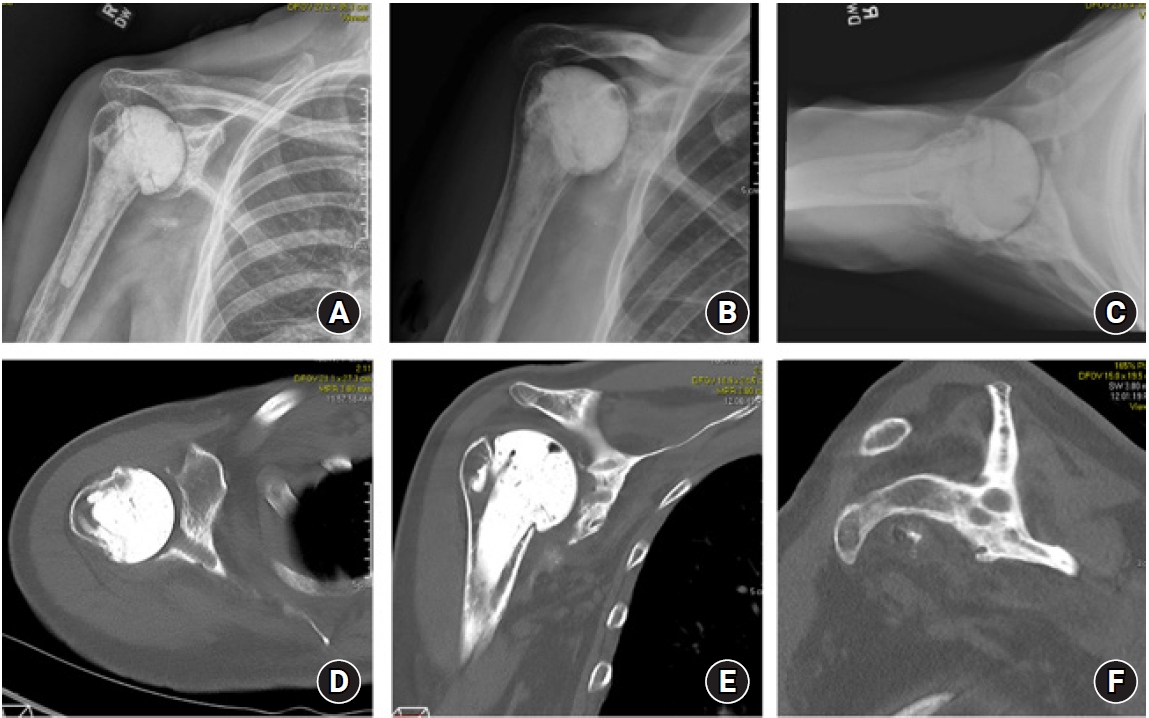
Fig. 2.

Fig. 3.
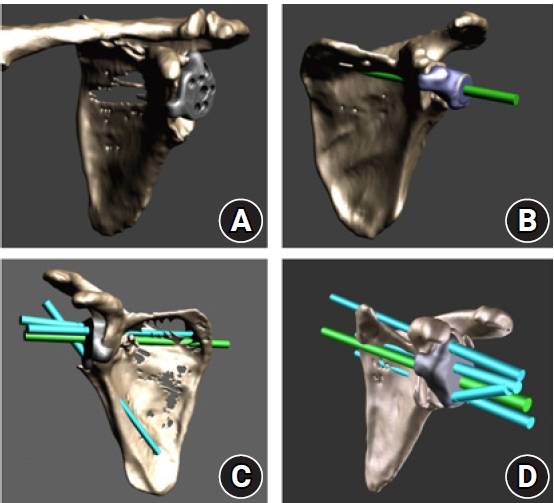
Table 1.
| Study | System used (manufacturer) | Setting | N |
No. (%) |
Mean (range) follow-up (mo) | No. (%) and type of complications | |
|---|---|---|---|---|---|---|---|
| Primary | Revision | ||||||
| Dines et al. (2017) [7] | Glenoid vault reconstruction system (VRS; Zimmer Biomet) | 1 Surgeon | 2 | 0 | 2 (100) | 33 (18–48) | 0 |
| Debeer et al. (2019) [6] | Glenius Glenoid Reconstruction System (Materialise NV) | 5 Centers | 10 | 4 (40) | 6 (60) | 31 (15–44) | 1 (10%): Instability |
| Rangarajan et al. (2020) [8] | Glenoid vault reconstruction system (VRS; Zimmer Biomet) | 1 Surgeon | 19 | 9 (47) | 10 (52) | 18 (12–27) | 4 (21%): 1 Each of nondisplaced GT fracture, intraoperative cortical perforation, recurrent instability and hematoma, early periprosthetic infection |
| Bodendorfer et al. (2021) [5] | Glenoid vault reconstruction system (VRS; Zimmer Biomet) | 4 Surgeons in 3 centers | 12 | 7 (58) | 5 (42) | 30 (24–52) | 0 |
| Porcellini et al. (2021) [9] | ProMade (LimaCorporate) | 2 Centers | 6 | 2 (33) | 4 (67) | 32 (25–38) | 1 (16%): Partial dislocation |
Table 2.
Table 3.
| Outcome | Preoperative | Postoperative | P-value |
|---|---|---|---|
| Range of motion (º) | |||
| Abduction | 73.9±33.1 | 95.6±39.7 | 0.03† |
| Flexion | 73.9±33.1 | 100.0±40.3 | 0.03† |
| External rotation | 46.7±16.6 | 60.0±20.2 | 0.05 |
| External rotation (arm at side) | 10.6±23.8 | 20.0±21.7 | 0.24 |
| Internal rotation | 17±15.8 | 6.1±31.6 | 0.39 |
| Internal rotation (hand behind back)* | Buttock | L4/L5 | 0.02† |
| Functional outcome | |||
| ASES score | 26.6±22.6 | 68.1±26.6 | <0.01† |
| SST | 4.0±3.0 | 8.0±2.8 | 0.02† |
| SANE | 34.7±22.0 | 69.6±27.3 | <0.01† |
| WOOS | 32.7±22.2 | 64.1±27.3 | <0.01† |
| Pain (visual analog scale) | 8.2±1.3 | 1.5±2.3 | <0.01† |
| Report satisfaction | 1.6 (1–2) | 3.7 (2–5) | <0.01† |
Values are presented as mean±standard deviation or median (interquartile range).
RTSA: reverse total shoulder arthroplasty, ASES: American Shoulder and Elbow Surgeons, SST: Simple Shoulder Test, SANE: Single Assessment Numeric Evaluation, WOOS: Western Ontario Osteoarthritis of the Shoulder Index.
REFERENCES
- TOOLS
-
METRICS

-
- 0 Crossref
- 1,577 View
- 30 Download
- Related articles in Clin Should Elbow
-
Postoperative pain control for shoulder arthroplasty ;0()
Patient-specific implants in reverse shoulder arthroplasty2024 March;27(1)
Variability in physical therapy protocols following total shoulder arthroplasty2023 September;26(3)







